Surgery


The Surgery team supports a wide range of projects across multiple programs at the Allen Institute. The team offers a variety of aseptic rodent surgical procedures ranging from stereotaxic injections to headpost implantation and cranial windowing. The ambitious scientific goals of the Neural Dynamics accelerator at Allen Institute involve integration of brain-wide circuit and cell-type mapping, complex optical and electrophysiological techniques, and various behavioral tasks. These projects require surgical preparations that provide the ability to precisely record from multiple brain regions across different behavioral states over extended periods of time. Through cross-team collaborations made possible by the Allen Institute’s unique structure and organization, the Surgery team has developed novel surgical approaches that provide unprecedented access to the brain while ensuring the highest standards of animal health and welfare. Combining decades of aseptic surgical experience with industry-inspired operating principles and data tracking, the Surgery team has established a state-of-the-art platform for driving modern systems neuroscience research at scale.

The Surgery team is tasked with performing large numbers of rodent surgeries in support of a variety of projects performed over many months to years. Performing surgeries at this scale requires a large team of well-trained surgeons who have developed expertise in procedures ranging from simple to complex. Standardization is achieved through specialized custom tooling, standard operating procedures (SOPs), and rigorous training processes. The team maintains its high level of surgical quality by relying on comprehensive quality control and quality assurance measures, which are tracked and analyzed for continued process improvement.

In addition to providing SOP-based surgeries, the Surgery team is critically involved in ongoing technical development. Through tight collaborations with Scientists, Engineers, and Veterinary staff, the Surgery team is continually developing and refining surgical preparations in support of the evolving scientific goals of the Allen Institute. Procedures are piloted, iterated on, and streamlined, resulting in standardized protocols that are publicly shared via protocols.io. In many cases, surgical development requires designing new surgical tooling and/or equipment, designs for which can be freely downloaded via our Hardware website.
In addition to training its staff, the Surgery team works with scientific staff from other departments to provide surgical training, certification, and support. The team also collaborates with, and in some cases trains, surgeons from external institutions through virtual and in-person interactions. Dissemination of surgical techniques and protocols is achieved through protocols.io, publications in peer-reviewed journals, as well as posters and presentations at conferences.

This platform builds on turn-key lightsheet microscopes to image mouse brains at high resolution and throughput. Analysis of the resulting volumetric images, including registration to standard brain coordinates and segmentation and counting of individual neurons, is fully automated.
This platform uses a two-photon microscope SLAP2 that records patterns of synaptic input and output in individual neurons at hundreds to thousands of frames per second in mice performing complex behaviors, using a flexible scan system that combines a digital micromirror device (DMD) with a high-speed scanner.
The Scientific Instrumentation and Process Engineering (SIPE) team is a shared engineering resource within the Allen Institute, focused on enabling and scaling cutting-edge bioscience through integrated hardware and software systems.
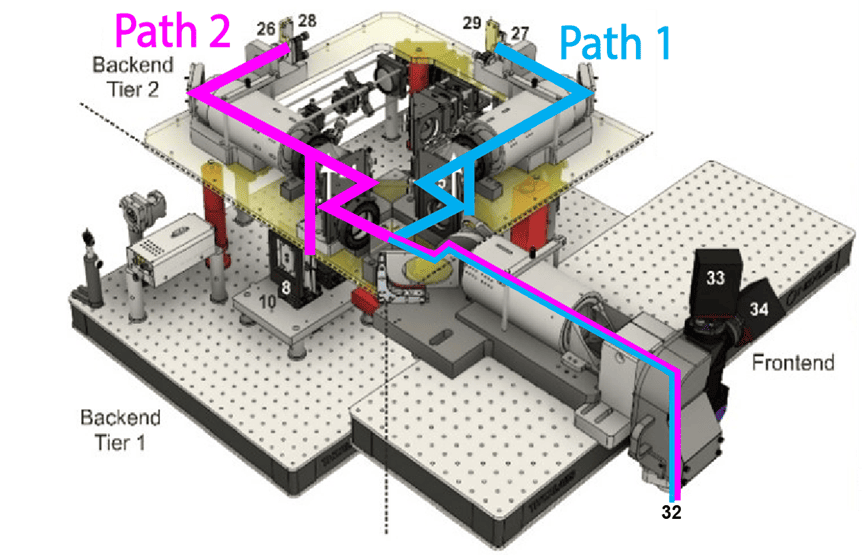
This team performs a variety of surgical procedures, including stereotaxic injections and implanting chronic cranial windows and Neuropixels probes.
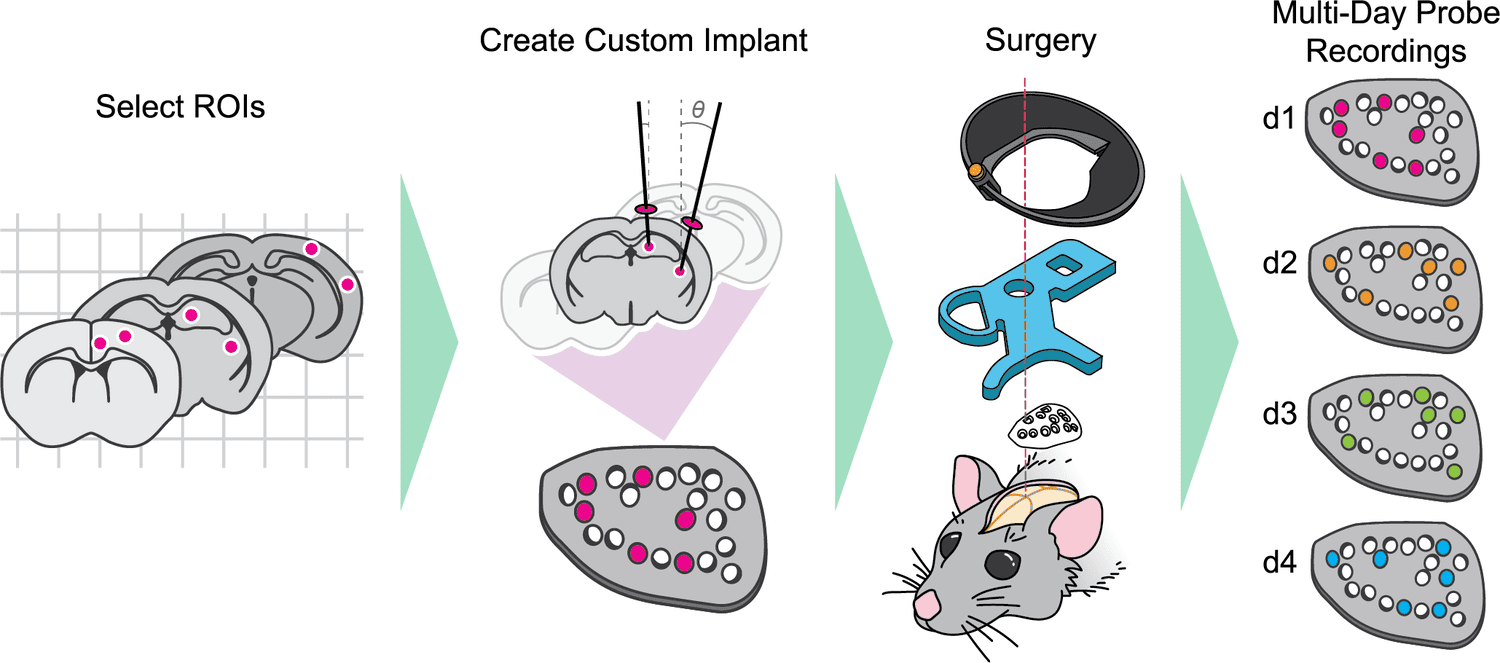
This platform implements pioneering technology for highly reproducible, targeted, brain-wide, cell-type-specific electrophysiology to record neural activity from defined neuron types across the brain. Analysis and quality control of the electrophysiology data are fully automated.
This platform combines innovative histology, ExA-SPIM microscopy, image handling, and machine learning to map the morphology and molecular identity of individual neurons across the whole brain at high throughput.
This platform enables optical measurement of neural activity and neurotransmitter release in populations of neurons to study neural circuit dynamics in behaving animals.
The Behavior platform uses advanced technology to implement a standardized, modular, multi-task virtual reality gymnasium for mice, with the goal to study brain function across different behaviors at scale.